您所在的位置:
首页
联系我们 |网站地图 |建议投诉 |版权声明 |使用帮助 |隐私安全
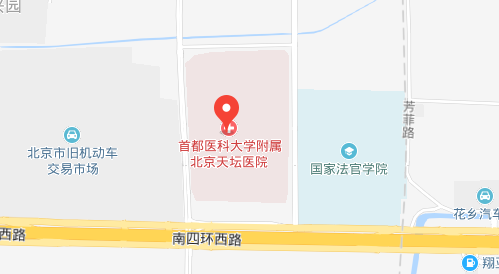
地址:北京市丰台区南四环西路119号 100070 电话:010-59976611
京ICP备05033471号-1 京卫网审字[2013]第0026号 文保网安备案号:1101010011
 京公网安备111010602104056 工信部链接:https://beian.miit.gov.cn/
京公网安备111010602104056 工信部链接:https://beian.miit.gov.cn/
©Copyright 2021 Beijing TianTan Hospital.All rights Reserved